March 27, 2026 - 14 minutes read

Supply Chain in Pharmaceutical Industry: Top Challenges and How to Overcome Them
The pharmaceutical supply chain is a high-stakes industry where delays or failures directly compromise patient outcomes. In this environment, operational efficiency is an absolute necessity. However, recent data suggests a significant gap between demand and delivery. As of late 2025, more than 216 prescription medicines were in short supply, with this scarcity impacting 88% of primary care physicians.
For pharmaceutical manufacturers, distributors, and 3PLs, resilience is a regulatory and operational mandate and requires a shift toward a unified data strategy. Below, we examine challenges biopharmaceutical companies face and how integrated supply chain visibility software provides the transparency needed to manage them. By addressing these challenges with centralized data, organizations can protect both their bottom line and the patients who depend on their products.
What Makes the Pharmaceutical Supply Chain Uniquely Complex?
Pharmaceutical supply chain management is defined by a global, multi-tier network that requires precision from sourcing to clinical delivery. Unlike traditional logistics, the biopharmaceutical sector operates under a zero-fail mandate. Every node in the chain must align to ensure product integrity and patient safety. Several factors contribute to this unique complexity:
- Multi-Tier Global Sourcing: Raw materials and active pharmaceutical ingredients (APIs) often travel through multiple countries and handoffs before reaching a manufacturing facility.
- Strict Regulatory Requirements: Compliance with FDA and GMP standards is non-negotiable. Every movement must be documented to provide a clear audit trail.
- Cold Chain Sensitivity: A significant portion of modern medicine, particularly biologics, requires strict temperature control. Even a brief deviation can render a shipment useless.
- Serialization and Traceability Mandates: Regulatory bodies now require package-level tracking to prevent counterfeiting and ensure a secure chain of custody.
Why Does Pharma Require Higher Visibility Than Other Industries?
While visibility is an advantage in all industries, it is an absolute necessity in healthcare and pharma. The stakes of these supply chains goes beyond lost revenue – they often involve public health and legal accountability. Achieving a high level of control requires pharmaceutical supply chain visibility software that can support these core capabilities:
- End-to-End Tracking: Managers must see the location and status of inventory across every mile, from the supplier’s warehouse to the pharmacy.
- Cold Chain Logistics and Temperature Tracking: Real-time monitoring of environmental conditions is essential to protect the efficacy of temperature-sensitive drugs.
- Compliance Documentation: Digital systems must automatically capture and store the data required for DSCSA and other regulatory frameworks, reducing the risk of human error.
- Exception Alerts: High visibility allows teams to receive instant notifications when a shipment is delayed or a temperature threshold is breached, enabling immediate intervention.
What Are the Top 8 Pharmaceutical Supply Chain Challenges?
While global logistics is inherently unpredictable, the pharmaceutical industry faces a unique set of hurdles that demand increased oversight. Identifying and addressing these pharma supply chain challenges is essential for maintaining a steady flow of life-saving medications.
1. Regulatory Compliance & DSCSA Requirements
The Drug Supply Chain Security Act (DSCSA) remains a central focus as the industry moves into a period of active enforcement.By early 2026, the stabilization and exemption periods for manufacturers, wholesalers, and large dispensers have concluded. While the goal is a secure, package-level trace for every drug, the operational reality is defined by “exception handling”—the process of resolving mismatches between physical products and their digital data.
This requirement has turned the loading dock into a pivotal pressure point. If a shipment arrives but the corresponding Electronic Product Code Information Services (EPCIS) data is missing or incorrectly formatted, the recipient cannot legally accept that product. According to a 2025 survey from the Partnership for DSCSA Governance (PDG), while nearly all manufacturers are sending serialized data, only 72% of downstream dispensers report receiving it successfully. This discrepancy forces products into quarantine, stalling inventory and creating a significant burden as teams manually reconcile data with upstream partners.
For many organizations, the challenge is shifting from technical implementation to operational execution. Teams must manage constant exceptions, such as unreadable barcodes or data files that arrive hours after the truck has departed. Because DSCSA records are designed to be immutable to ensure compliance, correcting clerical errors requires extensive coordination between stakeholders. Without a comprehensive visibility and collaboration strategy, these data gaps can lead to costly product rejections and delays.
2. Cold Chain & Temperature-Sensitive Logistics
The physical integrity of many pharmaceutical products depends on precise conditions during transit. The growth of cell and gene therapies and mRNA treatments has introduced significant vaccine supply chain challenges that require precise environmental control. Maintaining a consistent climate from the manufacturing facility to the patient is a high-stakes process where a single deviation can render a shipment useless.
This operational pressure continues to grow as the industry shifts further toward large-molecule medicine. According to Precedence Research, the global biologics market reached $487 billion in 2025 and is projected to surpass $534 billion in 2026. Because these products consist of living organisms or complex proteins, they are highly sensitive to humidity and temperature. Without real-time visibility into these environmental factors, companies risk financial losses and the potential delivery of compromised medicine.
3. Counterfeit Risk & Global Serialization
Protecting the pharmaceutical chain of custody is a global priority as manufacturers manage distribution networks that span multiple jurisdictions. While the US focuses on DSCSA compliance, domestic companies selling abroad must simultaneously navigate a patchwork of international serialization standards. This global serialization requirement means that a single product line may need to comply with the EU’s Falsified Medicines Directive (FMD), China’s tracking codes, and Brazil’s unique reporting laws, all while maintaining a secure audit trail.
Managing diverse mandates from a central hub is a substantial challenge. Without a unified visibility platform, companies must manage compliance in silos, increasing the risk of data errors and product delays at international borders. By centralizing serialization data, biopharma leaders can ensure that every package is accounted for and verified regardless of its destination. This oversight is the primary defense against the entry of counterfeit or diverted medications into the legitimate supply chain.
4. Supplier & Active Pharmaceutical Ingredient (API) Dependency
Over-reliance on concentrated geographic regions for active pharmaceutical ingredient supply chains has created an additional vulnerability in global health infrastructure. A high percentage of generic drug manufacturing sites are located in specific regions of Asia, meaning a single local disruption can trigger a global shortage. Data from the USP Medicine Supply Map highlights this concentration, noting that India and China together account for roughly 41% of the manufacturing sites that provide APIs for the U.S. market.
Geographic diversification is a long-term strategic goal for many biopharma leaders, but the immediate obstacle is managing the supplier risk within existing networks. Without visibility into the lower tiers of the supplier base, manufacturers are often unaware of a disruption until it reaches their own production line. Strengthening these dependencies requires digital integration with supplier networks to provide early warnings of delays or capacity issues.
5. Inventory Volatility & The Bullwhip Effect
Pharmaceutical companies often struggle with the bullwhip effect, a phenomenon where small fluctuations in patient demand create large swings in inventory requirements as they move up the supply chain. In an industry with long manufacturing lead times and high production costs, these imbalances lead to either costly overstocking or critical drug shortages. Balancing inventory management with lean operations is a constant struggle for logistics managers.
Limited visibility into point-of-care consumption often exacerbates this volatility. When manufacturers cannot see actual usage at the pharmacy or hospital level, they must make production decisions based on outdated or incomplete information. Integrating real-time demand data with supply chain visibility tools allows for more accurate forecasting and a more responsive production schedule. This approach reduces the financial impact of excess inventory and helps ensure that critical medications remain available for the patients who need them most.
6. The Order-to-Cash (O2C) Integration Gap
Many biopharmaceutical companies struggle to manage the order-to-cash cycle effectively because they rely on an outsourced model for manufacturing and distribution. While contract manufacturers (CMOs) handle the physical shipping, the biopharma firm remains responsible for delivery verification and revenue recognition. This creates a disconnect between outsourced logistics and internal financial systems. Without a standardized data layer, firms often lack the granular visibility needed to determine exactly when a product was delivered.
This deficit prevents teams from capturing essential documentation, such as proof of delivery, in real time. For many organizations, manual reconciliation processes lead to ongoing compliance challenges and delayed financial cycles. By centralizing logistics data and integrating it with internal ERP systems, organizations can automate revenue recognition at the line-item level. This connectivity can accelerate O2C cycles by several weeks, providing a significant advantage when meeting quarterly financial deadlines.
7. Global Disruptions & Evolving Trade Regulations
While international trade lanes are a constant concern, domestic disruptions in the U.S. have become a primary hurdle for pharmaceutical supply chains. Labor negotiations at major U.S. ports and shifting regional regulations create unpredictable lead times that ripple through the entire network. These delays are particularly impactful for medical device supply chain management, where precise delivery windows are often required for clinical procedures.
To mitigate these risks, companies are increasingly moving away from a reliance on single-carrier strategies. However, managing a multimodal network requires a high level of data agility. Without real-time visibility into carrier performance and port congestion, manufacturers cannot pivot effectively during a localized crisis. Strengthening resilience requires a data strategy that identifies bottlenecks before they result in stockouts, allowing teams to reroute shipments or adjust production schedules based on real-world conditions.
8. Data Silos & Fragmented Visibility
The most persistent challenge in the industry is the fragmentation of data across the pharmaceutical ecosystem. Most companies operate in a “black box” environment where critical information is trapped in disparate systems owned by 3PLs, carriers, and suppliers. This lack of connectivity makes it nearly impossible to achieve a unified operational view. While many organizations still rely on legacy EDI vs API in supply chain communication, these systems often lack the speed and granularity required for modern pharmaceutical visibility.
The solution lies in breaking down these silos to create a single source of truth for all logistics and compliance data. This connectivity is the foundation for every other operational improvement, from DSCSA compliance to O2C acceleration. Without a centralized platform to ingest and standardize data from every partner, organizations will continue to struggle with manual workflows and the high costs of operational invisibility.
How Can Supply Chain Visibility Improve Pharma Resilience?
Resilience in the pharmaceutical industry depends on the ability to transform passive data into proactive operational control. Visibility enables organizations to monitor shipments in real time, mitigate risk, and maintain constant compliance readiness. By centralizing data from every carrier, supplier, and distributor, biopharma leaders can identify potential disruptions before they impact patient care. This shift to a predictive model is essential for navigating future challenges in supply chain management, such as shifting trade policies and increasing logistics volatility.
The move toward digital transformation is already well underway. According to a PwC Digital Supply Chain Survey, 98% of pharmaceutical leaders state that digital tools have improved visibility into their end-to-end operations. This high adoption rate reflects a growing consensus that manual tracking is no longer sufficient for modern complexity. A digitally enabled supply chain provides the foundation for several critical improvements:
- Predictive Risk Mitigation: Using real-time data to identify weather or labor-related delays and rerouting shipments immediately.
- Automated Compliance: Capturing digital signatures and environmental data automatically to satisfy audits.
- Multimodal Flexibility: Managing air, ocean, and road freight through a single interface to maintain agility during regional crises.

How Does Unified Data Connect the Pharmaceutical Supply Chain?
A unified data layer transforms fragmented shipping updates into a coordinated operational workflow. Rather than replacing existing ERP or TMS applications, this central hub standardizes data from carriers, 3PLs, and CMOs. This ensures that every physical movement is mirrored by its digital twin in real time.
The modern pharmaceutical visibility workflow follows four critical stages:
- Data Ingestion and Standardization: The platform pulls and cleanses information from disparate sources, including ERP systems and carrier networks via API or EDI.
- Real-Time Milestone Tracking: Automated tracking provides real-time visibility into location, temperature status, and geofencing to prevent theft or diversion.
- Automated Exception Management: Predictive analytics flag potential issues, such as cold-chain breaches or transit delays, triggering immediate alerts for intervention.
- Compliance and Performance Intelligence: The system automatically captures DSCSA documentation and proof of delivery, feeding centralized dashboards that analyze carrier performance and landed costs.
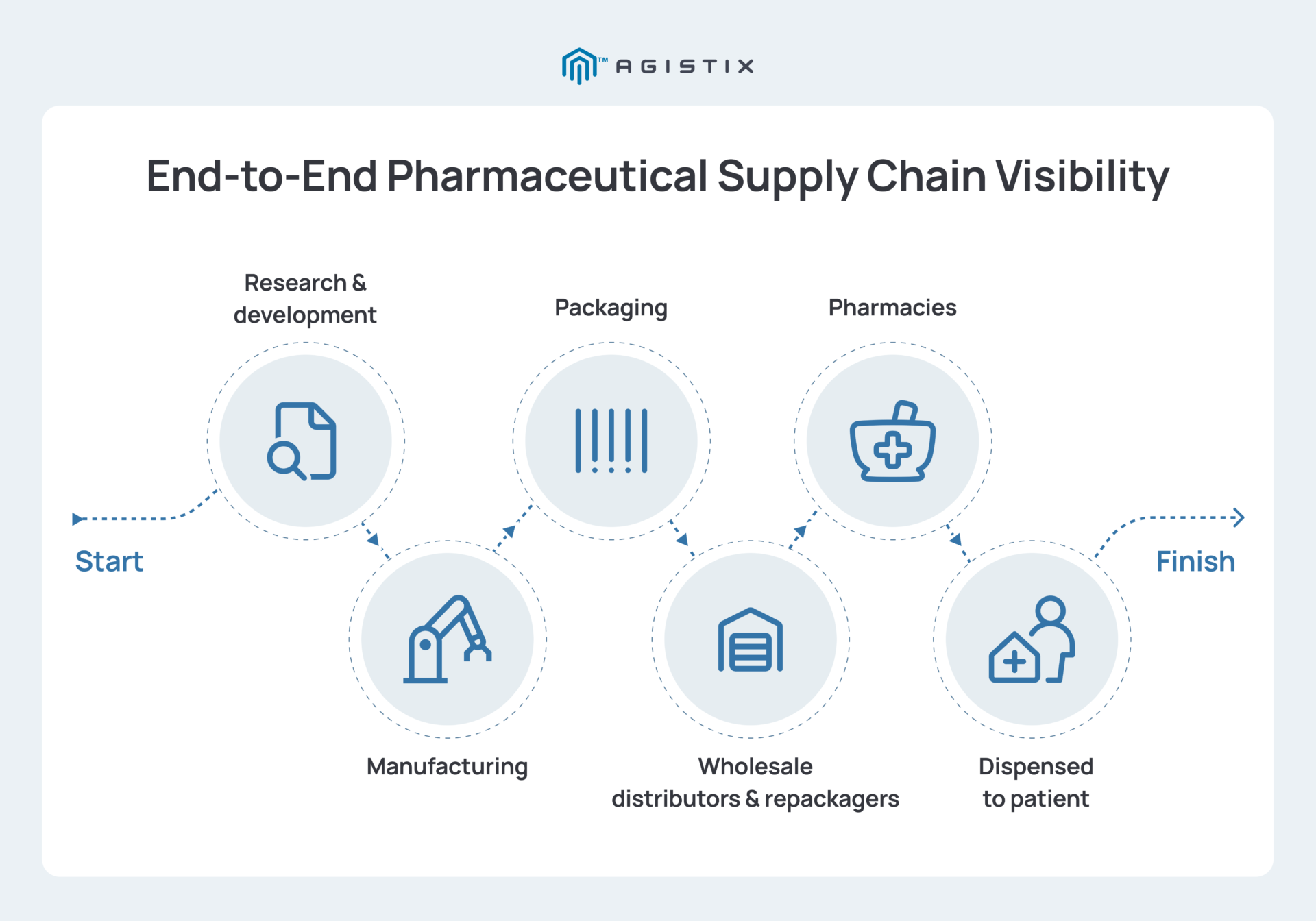
Best Practices to Strengthen Pharmaceutical Supply Chain Management
Building a resilient supply chain in the pharmaceutical industry requires shifting from manual oversight to an integrated, data-driven strategy. Firms can overcome pharma supply chain challenges and maintain compliance by adopting these best practices:
- Diversify Geographic Sourcing: Reducing dependency on concentrated API manufacturing regions prevents large-scale shortages and minimizes the impact of localized geopolitical disruptions.
- Transition to Actionable Intelligence: Traditional track-and-trace systems are reactive. Leading firms use serialization data to optimize product flow and identify delays before they result in a stockout.
- Invest in Digital Integration: Moving away from manual entry is essential. Organizations that invest in biopharmaceutical supply chain automation use data ingested from carriers and 3PLs to minimize errors and focus on exception management.
- Strengthen Collaboration: A single source of truth into shipment status allows for better coordination with 3PLs and CMO partners and faster response when issues arise.
- Adopt a Centralized Visibility Platform: Data silos cause operational delays. A centralized platform integrates existing ERP and TMS systems to provide the transparency needed for DSCSA compliance and O2C acceleration.
- Prioritize Predictive Analytics: Using historical performance data to forecast carrier reliability allows for more accurate inventory planning. This balances lean operations with the need for robust safety stock.
Strengthen Your Biopharma Supply Chain with Agistix
Managing the complexities of modern pharma logistics requires a level of data precision that manual processes cannot match. To protect patient outcomes and your bottom line, you need a unified visibility strategy that connects every stakeholder in real time.
Agistix provides the automation and intelligence required to simplify DSCSA compliance, accelerate revenue recognition, and safeguard your global distribution network.
Ready to transform your supply chain data into a competitive advantage? Schedule a demo of the Agistix platform today.
FAQ
What is the pharmaceutical supply chain?
The pharmaceutical supply chain is a highly regulated global network responsible for moving medications from raw material suppliers to patients. It involves a complex sequence of active pharmaceutical ingredient (API) manufacturers, wholesalers, 3PLs, and clinical providers. Because product integrity is a matter of public health, this specific supply chain requires a “zero-fail” approach to security and environmental control.
How do vaccine supply chain challenges differ from standard pharmaceutical logistics?
Vaccine supply chain challenges differ primarily in the intensity of environmental requirements and the narrow window for distribution. While standard medications might require simple refrigeration, many modern vaccines – particularly mRNA-based treatments – demand cryogenic or ultra-low temperature environments. These products are often high-volume and high-priority, meaning a single breakdown in the cold chain can impact the health of thousands of patients simultaneously.
Why is cold chain logistics critical in pharma?
Cold chain logistics is critical in pharma because temperature deviations can alter the chemical structure of specialty medicines and biologics, rendering them ineffective or dangerous. These products are often composed of complex proteins or living cells that degrade rapidly if they fall outside a specific thermal range. Maintaining a stable environment is a clinical necessity; without it, manufacturers risk significant financial losses and the potential delivery of compromised medication to patients.
How does DSCSA impact pharmaceutical supply chain management?
The Drug Supply Chain Security Act (DSCSA) mandates a fully electronic, interoperable system to trace prescription drugs at the package level. By early 2026, this has shifted from a technical goal to an active enforcement requirement. If the digital data (EPCIS) for a shipment does not perfectly match the physical product at the loading dock, the product cannot legally enter the supply chain. This mandate has made real-time exception handling and data synchronization the top priority for logistics leaders.
What technology improves pharmaceutical supply chain visibility?
Visibility is best improved through a centralized data layer that integrates with existing ERP, TMS, and 3PL systems. Instead of relying on manual updates or siloed spreadsheets, companies use supply chain visibility software to ingest data from every partner via API or EDI. This technology provides the “single source of truth” needed for real-time tracking, automated compliance documentation, and predictive exception alerts.
How can pharma companies reduce disruption risks?
Pharma companies can reduce disruption risks by implementing a diversified, data-driven strategy that prioritizes supply chain agility. Rather than relying on static logistics plans, resilient organizations use real-time visibility to identify and bypass bottlenecks before they impact delivery. Successful risk mitigation typically focuses on three core areas:
- Geographic Diversification: Reducing over-reliance on a single region for API sourcing to prevent localized crises from causing global shortages.
- Agile Multimodal Strategies: Using real-time visibility to pivot between air, ocean, and road freight when domestic or international trade lanes are blocked.
- Predictive Performance Monitoring: Analyzing historical carrier data to identify reliable partners and avoid predictable bottlenecks before they occur.

