
Case Study
A Biotech Case Study for Order-to-Cash Optimization
Company Overview
A global pharmaceutical company with more than $2.8 billion in annual revenue and operations across the U.S., Latin America, Europe, and Asia. The company develops targeted therapies to improve the quality of life for patients with cancer and other chronic conditions.
10 days faster order-to-cash cycle on average
FAP integration simplified GL coding & invoice reconciliation
40+ hours saved every month with automated document generation
The Challenge
The company’s global supply chain was expanding, but its core logistics processes remained fragmented and manual. Teams relied heavily on spreadsheets and lacked the ability to track supplier-booked freight or address delays in real time – an issue that posed a serious risk to temperature-sensitive shipments. International shipping was especially challenging , with slow, inconsistent documentation and no standardized workflows.
These gaps created several operational and compliance challenges:
- Disconnected systems for tracking, rating, and managing shipments across modes
- Limited visibility into in-transit shipments managed by suppliers and forwarders
- Inconsistent milestone updates impeded the ability to proactively manage delays
- No centralized commodity library for managing import and export details
- Manual customs documentation led to frequent mistakes and compliance issues
- Lacked automation to verify carrier invoices and manage GL codes quickly
- Inability to efficiently submit or reconcile proof-of-delivery data for invoicing and revenue recognition
The Agistix Solution
Agistix connected the client’s disjointed systems with a centralized logistics platform and supported end-to-end implementation across teams, carriers, and external users. The platform helped standardize workflows, improve data accuracy, and reduce risk across critical execution and documentation processes.
Shipment Visibility & Exception Management
- Enabled teams to monitor all shipments, including those booked by suppliers and forwarders, with rules-based alerts tied to delivery milestones and product type
- Improved response time and issue resolution for high-value, time-sensitive shipments through proactive exception alerts
Custom Documentation & Regulatory Compliance
- Automated DPL screening and generation of customs documents based on shipment attributes, reducing manual workload and improving accuracy
- Saved hours per week in manual document prep while ensuring compliance with FDA, USDA, and global customs requirements
Freight Audit & ERP Integration
- Connected carrier milestone data and invoice details to freight audit and ERP systems to streamline validation, automate GL code matching, and support timely revenue recognition
- Shortened the order-to-cash cycle by submitting PODs directly into ERP systems
Microsites for External Collaboration
- Deployed branded tools that allowed non-Agistix users—suppliers, warehouses, and forwarders—to initiate, track, and update shipments through controlled access points
- Extended platform capabilities without additional logins or training, improving collaboration and standardizing workflows
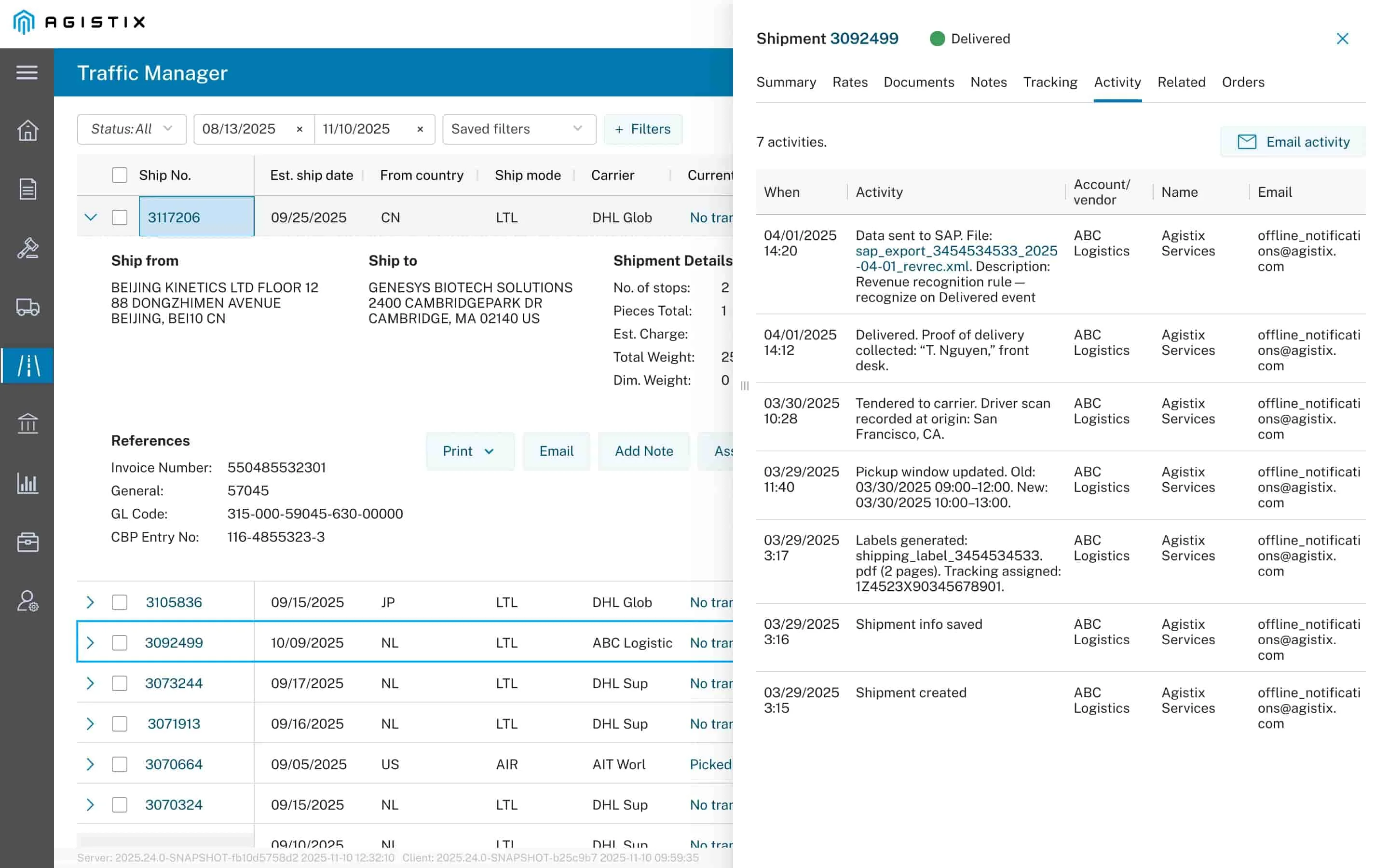
Results in Action
The client gained control over high-value international shipments, reduced revenue cycle delays, and eliminated manual processes through strategic collaboration and automation:
- Established carrier milestone requirements to monitor performance and ensure timely communication for critical, temperature-sensitive shipments
- Automated exception flags for late or missing milestones — including FDA, USDA, and Customs holds — enabling faster intervention and reduced risk
- Integrated freight audit and payment (FAP) to streamline GL coding
- Integrated proof-of-delivery data with Oracle and SAP to accelerate order-to-cash cycle by weeks
- Eliminated manual preparation of FDA use letters through automated document generation tied to shipment attributes
- Tagged priority shipments by product name to ensure added visibility and care for high-value or patient-sensitive deliveries
- Supported a 2-year ERP transition from Oracle to SAP by centralizing carrier integrations and standardizing milestone data across all modes and partners
What’s Next
- Automate ITN generation through AES integration
- Incorporate temperature sensor data into shipment tracking
- A QR codes to documents to strengthen chain-of-custody visibility
- Centralize all shipping and customs documentation within Agistix as the system of record
Building on a successful rollout, the client plans to expand their use of Agistix to:
Read More Case Studies

Case Study: Transforming Logistics Oversight and Trade Compliance

Case Study: Streamlining Cold-Chain Execution in Healthcare Diagnostics
